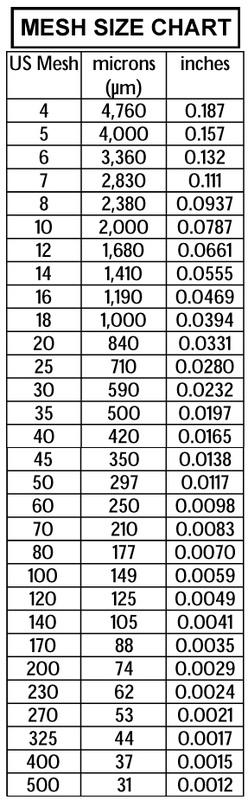
resource: Cameron Carbon Incorporated
In order to explain the capabilities of activated carbon an appreciation of its structure is most useful.
Much of the literature quotes a modified graphite-like structure; the modification resulting from the presence of microcrystallines, formed during the carbonization process, which during activation have their regular bonding, disrupted causing free valences which are very reactive. In addition, the presence of impurities and process conditions influence the formation of interior vacancies, in the microcrystalline structures.
Much of the literature quotes a modified graphite-like structure; the modification resulting from the presence of microcrystallines, formed during the carbonization process, which during activation have their regular bonding, disrupted causing free valences which are very reactive. In addition, the presence of impurities and process conditions influence the formation of interior vacancies, in the microcrystalline structures.
Such theory generally explains pores as the result of faults in crystalline structures.
However, more recent research studies provide a more feasible explanation of the carbon structure.
The generally accepted graphite-like structure theory falls down since the hardness of activated carbon is not in keeping with the layered structure of graphite. Furthermore, the manufacturing conditions are different; in particular the temperature range utilized for activated carbon production is lower than that required for graphitization.
Supporters of the graphite-like structure generally only explain the modified microcrystalline structure and ignore photographic and other methods of examining the residual macro structure.
High magnification electron scanning microscopy, at 20,000x magnification, has revealed the presence of residual cellular structures. These were previously unseen and unsuspected, except in the case of wood based activates which have sufficiently open structures visible to the naked eye.
Cellular units are built from sugars, the most important being glucose. Sugars ultimately will build to cellulose (the most important single unit in cellular construction) and cellulose polymers cross-link to form the wall of individual plant cells. Glucose units are wound into very tight helical spirals and under polarized light these exhibit anisotropy - demonstrating the presence of crystalline structures.
Although not as yet proven, it has been postulated that in the areas of maximum strain in cellulose chains it is conceivable that smaller crystalline units could be produced.
In addition to cellulose, other materials also exist in cell wall structure. Hemi-cellulose, which undergoes degradation more easily than cellulose and Lignin (the structure of which is still unproven) also exists and this is the most resistant to oxidation.
Most theories attribute the structure of activated carbon to be aromatic in origin, thus, allowing the carbon structure itself to be described as aromatic in order to explain active centers, etc. Structures of the size of cell dimensions obviously do not influence physical adsorption but illustrate that the only material available for oxidation lies within the cell walls themselves.
Final activates consist almost entirely of elemental carbon together with residual ash which, in the case of wood and coconut, originate from minerals within the vessels of living tissues; silica being the only constituent actually incorporated within the cell wall tissue matrix. The ash content of coal is of different composition and due to intrusion of inorganic materials during calcification.
Thus, the overall structure consists of a modified cellular-like configuration with varying ash components depending on the particular raw material.
The cellular-like structure theory offers a logical explanation for the differences in apparent density between activates of wood, coal and coconut.
Wood activates have a very open structure with thin wall cells whereas coconut activates show very thick walls with many pits.
Furthermore, measurements taken from photomicrographs of coconut show good agreement with mercury penetration data. It is known that the carbonization and activation processes destroy, to varying degrees, intercellular walls and sieve plates between cells. The end result on wood is a very open, sponge-like macrostructure seriously reducing the probability of adsorbate contact with cell walls. Activation of coconut produces a composition of rod-like cells in very close contact and large surface cavities are formed by destruction of dividing walls but these are shallow and do not extend through the activates granule.
The coconut activates thus differ significantly from wood activates in mechanical strength and density. Coconut activates exhibit extensive microspore volume, whereas wood activates have a definite trend to mesopores / macrospores and a corresponding change in their basic properties.
In the case of coal based carbons, pre-treatment of the raw coal is necessary in order for it to be processed, since raw coal swells during heating to produce coke-like structures. Control of this is achieved by first grinding the raw coal and mixing it with various additives, such as pitch, before it is introduced to the activation furnace. However, the grinding process destroys the mechanical strength of coal - therefore, ground coal is reconstituted into briquettes prior to processing.
Despite such pre-treatment, mercury penetration data for coal activates support the presence of structures similar to those identified in activates of wood and coconut, but to date no detection of residual plant structures has been found in coal activates. Isotherm determinations reveal extensive microspore structures, although coal activates’ pore spectra are different to those of coconut activates with a tendency toward macrospores at lower activation.
The most reliable carbon structure model suggested to date is similar to that of polyamantane (C66 H59) which allows for a large degree of non-aromaticity, electron transfer and resonance. Progressive activation would tend to increase the number of active sites, and in turn the surface activity, similar to observed reactions with higher activates.
However, more recent research studies provide a more feasible explanation of the carbon structure.
The generally accepted graphite-like structure theory falls down since the hardness of activated carbon is not in keeping with the layered structure of graphite. Furthermore, the manufacturing conditions are different; in particular the temperature range utilized for activated carbon production is lower than that required for graphitization.
Supporters of the graphite-like structure generally only explain the modified microcrystalline structure and ignore photographic and other methods of examining the residual macro structure.
High magnification electron scanning microscopy, at 20,000x magnification, has revealed the presence of residual cellular structures. These were previously unseen and unsuspected, except in the case of wood based activates which have sufficiently open structures visible to the naked eye.
Cellular units are built from sugars, the most important being glucose. Sugars ultimately will build to cellulose (the most important single unit in cellular construction) and cellulose polymers cross-link to form the wall of individual plant cells. Glucose units are wound into very tight helical spirals and under polarized light these exhibit anisotropy - demonstrating the presence of crystalline structures.
Although not as yet proven, it has been postulated that in the areas of maximum strain in cellulose chains it is conceivable that smaller crystalline units could be produced.
In addition to cellulose, other materials also exist in cell wall structure. Hemi-cellulose, which undergoes degradation more easily than cellulose and Lignin (the structure of which is still unproven) also exists and this is the most resistant to oxidation.
Most theories attribute the structure of activated carbon to be aromatic in origin, thus, allowing the carbon structure itself to be described as aromatic in order to explain active centers, etc. Structures of the size of cell dimensions obviously do not influence physical adsorption but illustrate that the only material available for oxidation lies within the cell walls themselves.
Final activates consist almost entirely of elemental carbon together with residual ash which, in the case of wood and coconut, originate from minerals within the vessels of living tissues; silica being the only constituent actually incorporated within the cell wall tissue matrix. The ash content of coal is of different composition and due to intrusion of inorganic materials during calcification.
Thus, the overall structure consists of a modified cellular-like configuration with varying ash components depending on the particular raw material.
The cellular-like structure theory offers a logical explanation for the differences in apparent density between activates of wood, coal and coconut.
Wood activates have a very open structure with thin wall cells whereas coconut activates show very thick walls with many pits.
Furthermore, measurements taken from photomicrographs of coconut show good agreement with mercury penetration data. It is known that the carbonization and activation processes destroy, to varying degrees, intercellular walls and sieve plates between cells. The end result on wood is a very open, sponge-like macrostructure seriously reducing the probability of adsorbate contact with cell walls. Activation of coconut produces a composition of rod-like cells in very close contact and large surface cavities are formed by destruction of dividing walls but these are shallow and do not extend through the activates granule.
The coconut activates thus differ significantly from wood activates in mechanical strength and density. Coconut activates exhibit extensive microspore volume, whereas wood activates have a definite trend to mesopores / macrospores and a corresponding change in their basic properties.
In the case of coal based carbons, pre-treatment of the raw coal is necessary in order for it to be processed, since raw coal swells during heating to produce coke-like structures. Control of this is achieved by first grinding the raw coal and mixing it with various additives, such as pitch, before it is introduced to the activation furnace. However, the grinding process destroys the mechanical strength of coal - therefore, ground coal is reconstituted into briquettes prior to processing.
Despite such pre-treatment, mercury penetration data for coal activates support the presence of structures similar to those identified in activates of wood and coconut, but to date no detection of residual plant structures has been found in coal activates. Isotherm determinations reveal extensive microspore structures, although coal activates’ pore spectra are different to those of coconut activates with a tendency toward macrospores at lower activation.
The most reliable carbon structure model suggested to date is similar to that of polyamantane (C66 H59) which allows for a large degree of non-aromaticity, electron transfer and resonance. Progressive activation would tend to increase the number of active sites, and in turn the surface activity, similar to observed reactions with higher activates.